 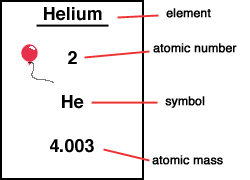
For hydrogen, the atomic mass is 1 because there is one proton and no neutrons. The number of protons is the atomic number, and the number of protons plus neutrons is the atomic mass. The positively charged protons tend to repel each other, and the neutrons help to hold the nucleus together. All other elements have neutrons as well as protons in their nucleus, such as helium, which is depicted in Figure 2.2. The proton forms the nucleus, while the electron orbits around it. The element hydrogen has the simplest atoms, each with just one proton and one electron. Table 2.1 Charges and masses of the particles within atoms Elementary Particle Both protons and neutrons have a mass of 1, while electrons have almost no mass. The negative charge of one electron balances the positive charge of one proton. As summarized in Table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Chem.2.1 Electrons, Protons, Neutrons, and AtomsĪll matter, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: protons, neutrons, and electrons. SOURCE Atomic weights of the elements: Review 2000 by John R de Laeter et al. Studies that have contributed decisively to our understanding of quantum physics and chemistry In gas, liquid, and solid phases and of 3He and 4He mixtures have been the subjects of many experimental With minor amounts derived from beta decay of 3H, which is produced by the reaction 6Li(n,α) 3H in the earth, by cosmic-ray reactions in the atmosphere, or by nuclear explosions and industry.Īccumulations of 3He produced by radioactive decay of anthropogenic 3H provide a useful tool for determiningĪges of groundwaters and surface waters, including the ocean. The minor isotope 3He is believed to be largely primordial, Produced by alpha decay of heavy radioisotopes. Helium is too light toīe held in the atmosphere by Earth's gravitation over periods comparable with the age of the earth thus,ĤHe in the atmosphere has been derived almost entirely from degassing of the solid earth, where it is 
In voids in clathrate compounds, He occurs naturally only as a monoatomic gas. Apart from its presence in gaseous or fluid inclusions and interstitial positions in crystals, and Helium, a noble gas, is chemically the most unreactive element with the lowest boiling point (4.2 K). Those types of 3He-enriched sources are considered to represent emissions of primordial heliumįrom incompletely degassed regions deep within the earth. Some of which have 3He abundances more than ten times that of atmospheric helium, hence the annotation Sources, but it does not include all helium from volcanic rocks or associated geothermal springs and gases, This interval includes some natural 3He-enriched The indicated interval for the standard atomic weight of He has a lower limit of 4.002 600, corresponding In 1969 the Commission recommended A r(He) = 4.002 60(1), which was identical to the atomic mass of 4He to six significant figures.Ī subsequent determination of the isotopic composition of atmospheric helium have confirmed this value which has been last revised in 1983. Is present in natural sources of helium with a smaller abundance than that of any other stable isotope relative Of 0.000 137 % as determined by Nier had a negligible effect on the atomic weight of helium. The atomic mass of 4He to four decimal places. In its 1961 report, the Commission recommended A r(He) = 4.0026 based on 
In contrast to the other noble gases, which are separated for commercial use almost entirely fromĪir, a large amount of commercial He is derived from natural gas deposits containing relatively high concentrations
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
